Topic

Targeting cholangiocarcinoma cells by cold piezoelectric plasmas : in vitro efficacy and cellular mechanisms
– Authors : M. Soulier, B. Lekbaby, I. Houari, H. Decauchy, A. Pavy, A. Coumes, R. Morichon, T. Dufour, L. Fouassier
– References : Scientific Reports, 14:30178 (2024)
– Links : DOI, HAL (free download)
Abstract
Cold piezoelectric plasma (CPP) is a novel approach in cancer therapy, enabling the development of portable treatment devices capable of triggering cancer cell death. While its effectiveness remains underexplored, this research focuses on its application against cholangiocarcinoma (CCA), an aggressive cancer of the biliary tract. A CPP device is utilized to generate either a corona discharge (Pz-CD) or a dielectric barrier discharge (Pz-DBD) for in vitro experiments. Notably, Pz-CD can deliver more power than Pz-DBD, although both sources produce significant levels of reactive species in plasma and liquid phases. This work shows that CPP causes a gradient increase in medium temperature from the center towards the edges of the culture well, especially for longer treatment times. Although Pz-CD heats more significantly, it cools quickly after plasma extinction. When applied to human CCA cells, CPP shows immediate and long-term effects, more localized for Pz-CD, while more uniform for Pz-DBD. Immediate effects result also in actin cytoskeleton remodeling without alteration of the cell membrane permeability. Long-term effects of CPP, although the antioxidant system is engaged, include activation of the DNA damage response pathway leading to cell death. In conclusion, CPP should be recognized as a promising antitumor therapy.

Les plasmas froids et le Vivant, de nouvelles avancées
– Authors : C. Douat, J. Santos Sousa, T. Dufour
– References : Reflets de la Physique, Vol. 75, pp. 24-30 (2023)
– Links : DOI, Free download
Abstract
Depuis le début des années 2000, des avancées technologiques majeures ont permis l’émergence de plasmas froids à pression atmosphérique ayant de faibles valeurs de courant et des températures proches de la température ambiante. Dans cet article, nous exposons dans un premier temps les principales sources de plasma froid utilisées avec succès dans les applications biomédicales, en insistant notamment sur les propriétés physico-chimiques recherchées. Dans un second temps, nous proposons un état de l’art des dernières avancées médicales (en particulier en cancérologie et en dermatologie), ainsi qu’en agriculture.
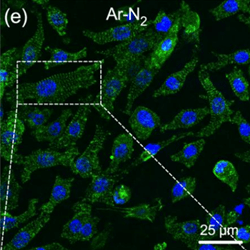
Cyto- and bio-compatibility assessment of plasma-treated polyvinylidene fluoride scaffolds for cardiac tissue engineering
– Authors : M. Kitsara, G. Revet, J.-S. Vartanian-Grimaldi, A. Simon, M. Minguy, A. Miche, V. Humblot, T. Dufour, O. Agbulut
– References : Front. Bioeng. Biotechnol., Vol. 4, pp. 18 (2022)
– Links : DOI, HAL (free download)
Abstract
As part of applications dealing with cardiovascular tissue engineering, drop-cast polyvinylidene fluoride (PVDF) scaffolds have been treated by cold plasma to enhance their adherence to cardiac cells. The scaffolds were treated in a dielectric barrier device where cold plasma was generated in a gaseous environment combining a carrier gas (helium or argon) with/without a reactive gas (molecular nitrogen). We show that an Ar-N2 plasma treatment of 10 min results in significant hydrophilization of the scaffolds, with contact angles as low as 52.4° instead of 132.2° for native PVDF scaffolds. Correlation between optical emission spectroscopy and X-ray photoelectron spectroscopy shows that OH radicals from the plasma phase can functionalize the surface scaffolds, resulting in improved wettability. For all plasma-treated PVDF scaffolds, the adhesion and maturation of primary cardiomyocytes is increased, showing a well-organized sarcomeric structure (α-actinin immunostaining). The efficacy of plasma treatment was also supported by real-time PCR analysis to demonstrate an increased expression of the genes related to adhesion and cardiomyocyte function. Finally, the biocompatibility of the PVDF scaffolds was studied in a cardiac environment, after implantation of acellular scaffolds on the surface of the heart of healthy mice. Seven and 28 days after implantation, no exuberant fibrosis and no multinucleated giant cells were visible in the grafted area, hence demonstrating the absence of foreign body reaction and the biocompatibility of these scaffolds.

Cold plasma endoscopy applied to biliary ducts : feasibility risk assessment on human-like and porcine models for the treatment of cholangiocarcinoma
– Authors : H. Decauchy, A. Pavy, M. Camus, L. Fouassier, T. Dufour
– References : J. Phys. D : Appl. Phys., Vol. 55, No. 45, 455401, 23pp (2022)
– Links : DOI, HAL (free download)
Abstract
For the past twelve years, cold plasmas (i.e., weakly ionized gas) have been positioned as a breakthrough technology for treating cancer thanks to their antitumor properties. The innovation of ad hoc plasma sources and personalized protocols appears crucial to treat cancers with a very poor prognosis. This is the case for cholangiocarcinoma (CCA), a biliary tract cancer, whose treatment with cold plasma is envisioned but requires the innovation of catheters and endoscopic devices for local therapies. Before conducting clinical trials, the performances and limitations of cold plasma endoscopy must be evaluated in terms of safety for both the patient and clinician as well as in terms of therapeutic efficacy. These objectives are pursued in the present work, in which a transferred plasma catheter is used, powered by 8-10 kV, 1-2 μs wide, 5-10 kHz repeated pulses, allowing guided streamers to be transferred over lengths of at least 2 m. In a first step, the catheter is utilized without the duodenoscope and directly inserted into an artificial model reproducing the topography and the electrical response of the biliary tree. This model allows to validate the technical feasibility of the technology and to demonstrate the absence of electrical and thermal risks. Indeed, the voltage and current deposited are as low as 3.98 V and 1.19 mA (RMS values) respectively, while the temperature locally increases from 23°C to 27 °C. In a second step, the catheter is inserted into the duodenoscope, the whole being applied to a porcine anatomical model. After passing through the esophagus, stomach and duodenum, the distal part of the duodenoscope is placed at the entrance to the papilla so that the catheter can easily enter the choledoc and then the common bile duct. Interestingly, the electrical power values deposited are of the order of 100 mW especially because the current values are at least 10 times higher. These more elevated values of electrical parameters but also electromagnetic effects are discussed considering physical aspects like eddy currents. The absence of electrical and thermal risks is demonstrated and consolidated by the IEC standards for medical devices. In a third time, we demonstrate that the cold plasma catheter can induce antitumor effects on in vitro experimental models of human CCA. The methodology proposed in this article validates the relevance of cold plasma endoscopy as a potential local treatment for cholangiocarcinoma and allows bridging cognitive and patient-oriented research.
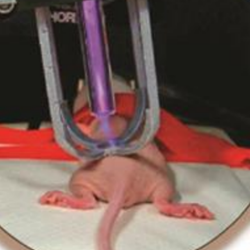
Cold atmospheric plasma induces tumor cell death in preclinical in vivo and in vitro models of human cholangiocarcinoma
– Authors : J. Vaquero, F. Judée, M. Vallette, H. Decauchy, A. Arbelaiz, L. Aoudjehane, O. Scatton, E. Gonzalez-Sanchez, F. Merabtene, J. Augustin, C. Housset, T. Dufour and Laura Fouassier
– References : Cancers, 12(5), 1280 (2020)
– Links : DOI, HAL (free download)
Abstract
Through the last decade, cold atmospheric plasma (CAP) has emerged as an innovative therapeutic option for cancer treatment. Recently, we have set up a potentially safe atmospheric pressure plasma jet device that displays antitumoral properties in a preclinical model of cholangiocarcinoma (CCA), a rare and very aggressive cancer emerging from the biliary tree with few efficient treatments. In the present study, we aimed at deciphering the molecular mechanisms underlying the antitumor effects of CAP towards CCA both in an in vivo and in vitro context. In vivo, using subcutaneous xenografts into immunocompromised mice, CAP treatment of CCA induced DNA lesions and tumor cell apoptosis, as evaluated by 8-oxoguanine and cleaved caspase-3 immunohistochemistry, respectively. Analysis of the tumor microenvironment showed changes in markers related to macrophage polarization. In vitro, incubation of CCA cells with CAP-treated culture media (i.e. plasma-activated media, PAM) led to a dose response decrease in cell survival. At molecular level, CAP treatment induced double-strand DNA breaks, followed by an increased phosphorylation and activation of the cell cycle master regulators CHK1 and p53, leading to cell cycle arrest and cell death by apoptosis. In conclusion, CAP is a novel therapeutic option to consider for CCA in the future.

Atmospheric pressure plasma jets applied to cancerology : correlating electrical configuration with in vivo toxicity and therapeutic efficiency
– Authors : F. Judée, J. Vaquero, L. Fouassier, S. Guegan, T. Dufour
– References : Journal of Physics D : Applied Physics, Vol. 52, Issue 24, 2019
– Links : DOI, HAL (free download)
Abstract
Two atmospheric pressure plasma jet (APPJ) devices—a plasma gun and a plasma Tesla jet—are compared in terms of safety and therapeutic efficiency to reduce the tumor volume progression of cholangiocarcinoma, i.e. a rare and very aggressive cancer emerging in biliary tree. For this, a three steps methodology is carried out. First, the two APPJ have been benchmarked in regard to their electrical and physico-chemical properties while interacting with material targets: dielectric plate, liquid sample, metal plate and an equivalent electrical circuit of human body. The propagation properties of the ionization wave interacting with these targets are discussed, in particular the profile of the related pulsed atmospheric plasma streams. In a second step, a dermal toxicity survey is performed so as to define an experimental operating window where plasma parameters can be changed without damaging healthy skin of mice during their exposure to plasma and without inducing any electrical hazards (burnings, ventricular fibrillation). Optimal conditions are identified discarding the conditions where slight alterations may be evidenced by histology (e.g. prenecrotic aspect of keratinocytes, alterations in the collagen structure). Hence, for the two APPJ plasma parameters these conditions are as follow: duty cycle = 14%, repetition frequency = 30 kHz, magnitude = 7 kV, gap = 10 mm and exposure time = 1 min. In a third step, the two plasma jets are utilized on cholangiocarcinoma xenograft tumor model developed in immunodeficient mice. The two devices are safe and a significant therapeutic efficiency is demonstrated with the plasma Tesla. In conclusion, we have developed a safe cold atmospheric plasma device with antitumoral properties in preclinical model of cholangiocarcinoma, opening the path for new anticancer treatment opportunities.
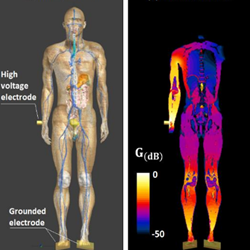
Plasma gun for medical applications : engineering an equivalent electrical target of human body and deciphering relevant electrical parameters
– Authors : F. Judée, T. Dufour
– References : Journal of Physics D : Applied Physics, Letter, Vol. 52, Issue 16, 2019
– Links : DOI, HAL (free download)
Abstract
Simulations and experimental works have been carried out in a complementary way to engineer a basic material target mimicking the same dielectric properties of the human body. It includes a resistor in parallel with a capacitor, whose values (Rh=1500 Ω and Ch=100 pF) are estimated in regard of parameters commonly utilized upon in vivo campaigns (frequency=30 kHz, gap=10 mm, high voltage electrode surface=12.6 mm2). This equivalent electrical human body (EEHB) circuit can be used as a reference and realistic target to calibrate electrical properties of therapeutic plasma sources before their utilization on patients. In this letter, we consider a configuration where this EEHB target interacts with a plasma gun (PG). Plasma power measurements performed in such configuration clearly indicate two operating modes depending on the value of the supplied voltage. Hence, the plasma gun generates pulsed atmospheric plasma streams likely to present therapeutic interest for voltages comprised between 3.0 and 8.5 kV while for higher values, transient arcs of thermal plasma are generated and represent substantial risks for the patient.